Modelling
Reporting, summarising and communicating models in R
Welcome back! 🏖
Last session we talked a bit about how you might scrape innovative data from the web. Collecting data is not an end in itself, rather you do it to be able to run models that explore, infer or predict outcomes. We won’t focus on the statistical considerations that inform your model choice, instead we will show you how to:
- use formulas to specify multiple models
- process (estimate) model results with
broom - summarise outputs with
modelsummary - communicate results through plots and tables
The Model Workflow with R
While the last session dealt with one of the sexier areas of Data Science (Web Scraping), today we will go back to the bread and butter 🍞 of any self-respecting data scientist - modelling!
But if we are not even talking about ML, DL or model choice, why bother? Well, as with most things it is worth to revisit fundamentals from time to time. By investing just a little more effort here, we can create much better reports and papers!
In general, remember, your basic workflow for evaluating and reporting models is the following:
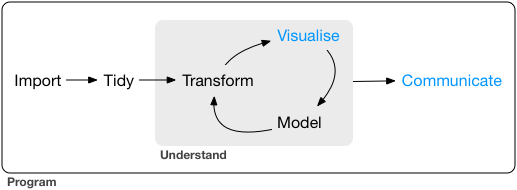
Today we will mostly deal with the model and communicate steps in the graph.
Setup
Before we get started with the actual coding we need to load a few packages and get some data. We are using this WHO dataset on life expectancy.
pacman::p_load(kableExtra, tidyverse, broom, modelsummary, specr, janitor, modelr, wesanderson)
life_expec_dat <- read_csv("life_expectancy.csv") %>%
janitor::clean_names() %>%
mutate(developed = if_else(status == "Developed", 1, 0),
large = if_else(population > 50000000, 1, 0))
head(life_expec_dat)Using Formulas in R 👩🏫
R as coding language was specifically designed with statistical
analysis in mind. Therefore the founding fathers of R 🙌 - in their
infinite wisdom - decided to create a special object class (called
formula) to help us run models.
Syntax
A quick recap on the syntax that you can use within formulas. The
~ sign generally differentiates between dependent and
independent variables. The + sign is used to distinguish
between independent variables. Here are a few other lines that are nice
to know:
y ~ x1 + x2 # standard formula
y ~ . # include all other columns in data as x vars
y ~ x1 * x2 # Interaction terms
y ~ x + I(x^2) # higher order termsformulas are very straightforward to work with. To avoid any errors you should follow the following two steps.
Step 1: create a string containing the written
formula. The paste function from last week is useful for
this.
formula_string <- paste("life_expectancy", "~ gdp")
formula_string## [1] "life_expectancy ~ gdp"Step 2: transform the string into R class formula. Only then can you plug it into the model function.
form <- as.formula(formula_string)# needs to be transformed to correct class
form## life_expectancy ~ gdpHere is the proof that both ways of doing it are identical.
form <- lm(form, data = life_expec_dat)
reg <- lm(life_expectancy ~ gdp, data = life_expec_dat)
reg2 <- glm(life_expectancy ~ gdp, family = "gaussian", data = life_expec_dat)
form$coefficients == reg$coefficients## (Intercept) gdp
## TRUE TRUEform$coefficients == reg2$coefficients## (Intercept) gdp
## TRUE TRUEFor Iteration
We encourage you to fit many models, ranging from simple to complex, and formulas can help you in the iteration process.
Often, the modelling process requires you to run the same specification with multiple configurations of both dependent and independent variables. Model formulas make running many similar models super easy:
Step 1: You define a function that let’s you plug in different variables at x or y. These functions take the y or x variable(s) as a string and return a model object.
# function to include different y variables.
lm_fun_iter_y <- function(dep_var) {
lm( as.formula(paste(dep_var, "~ gdp")), data = life_expec_dat)
}
# function to include different x variables.
lm_fun_iter_x <- function(indep_var) {
lm( as.formula(paste("life_expectancy ~", paste(indep_var, collapse = "+"))), data = life_expec_dat)
}Notice: It is unlikely you will run
just a simple bivariate regression (i.e. your model will include many
independent variables). Therefore, we need to use a nested paste
function that combines (collapse()) the input character
vector with +.
Step 2: You use map() (cf. lab-session
4 to iterate the model over a vector or list of input variables:
# create vector of variables to iterate over
vars <- life_expec_dat %>%
select(-c("life_expectancy", "country")) %>%
names()
# run a bivariate model for each column
biv_model_out <- vars %>%
map(lm_fun_iter_x)
biv_model_out## [[1]]
##
## Call:
## lm(formula = as.formula(paste("life_expectancy ~", paste(indep_var,
## collapse = "+"))), data = life_expec_dat)
##
## Coefficients:
## (Intercept) year
## -635.872 0.351
##
##
## [[2]]
##
## Call:
## lm(formula = as.formula(paste("life_expectancy ~", paste(indep_var,
## collapse = "+"))), data = life_expec_dat)
##
## Coefficients:
## (Intercept) statusDeveloping
## 79.2 -12.1
....This returns a list of all the models that your ran. Given that there
are a quite a few models now, so it might be an idea to retain the names
of the variables that you feed into your model. You can do this with
purrr::set_names().
# run a bivariate model for each column
biv_model_out_w_names <- vars %>%
set_names() %>%
map(lm_fun_iter_x)
biv_model_out_w_names$year ##
## Call:
## lm(formula = as.formula(paste("life_expectancy ~", paste(indep_var,
## collapse = "+"))), data = life_expec_dat)
##
## Coefficients:
## (Intercept) year
## -635.872 0.351But say we are interested to see what effect gdp has on life_expectancy, but you suspect that other covariates like alcohol consumption, health care expenditure, average bmi and the propagation of aids also make sense. And it is likely that multiple potential combinations factor into life_expectancy. Would it not be best if you were to run several different multivariate models? Let’s go over the code that you saw in the lecture:
Step 1: Find out how many possible combinations of independent and dependent variables exist:
combinations <-
map(1:5, function(x) {combn(1:5, x)}) %>% # create all possible combinations (we have 5 covariates)
map(ncol) %>% # extract number of combinations
unlist() %>% # pull out of list structure
sum() # compute sum
combinations## [1] 31Step 2: Write a function that can run all possible combinations in one go:
combn_models <- function(depvar, covars, data) {
combn_list <- list()
# generate list of covariate combinations
for (i in seq_along(covars)) {
combn_list[[i]] <- combn(covars, i, simplify = FALSE)
} # what is combn?
combn_list <-
unlist(combn_list, recursive = FALSE) # easier to not work with list object
# function to generate formulas
gen_formula <- function(covars, depvar) {
form <-
as.formula(paste0(depvar, " ~ ", paste0(covars, collapse = "+")))
form
}
# generate formulas
formulas_list <-
purrr::map(combn_list, gen_formula, depvar = depvar)
# run models
models_list <- purrr::map(formulas_list, lm, data = data)
models_list
}Step 3: Apply the function to our case:
depvar = "life_expectancy"
covars = c("gdp", "percentage_expenditure", "alcohol", "bmi", "hiv_aids")
multiv_model_out <- combn_models(depvar = depvar, covars = covars, data = life_expec_dat)
# Check that we have the correct number of models
length(multiv_model_out)## [1] 31As you can see the function in the lecture is quite powerful, but it is not the only option. Depending on the type of analysis that you plan to run certain packages have built-in options to run combinations of regression models in only a few lines (see for example the multiple estimations vignette for the fixest package).
Specification Curve Analysis ⤴️
In the above example, I made a conscious choice to run these specific models. This conscious choice should reflect insights from theory (here: not so much) and be defensible (eh…). Unfortunately, some researchers argue that by making a conscious choice, I select an arbitrary or possibly even biased model specification. They propose to approach model specification analytically.
They call this Specification Curve Analysis, or Multiverse Analysis.
There is a very nice package to run this: specr. In very
simplistic terms it follows this path:
First you compile a list of defensible “model ingredients”:
setup_specs(
y = c("life_expectancy"),
# We add an additional dependent variable
x = c("gdp", "log(gdp)", "I(gdp^2)", "gdp * total_expenditure"),
# We are not sure which independent variable is better
model = c("lm", "glm"),
# We only use linear model or more complicated one
controls = c("population", "alcohol", "bmi"))Second you define additional analytical choices such as: model family, potential subgroups to run it over etc.
# Run specification curve analysis (under the hood it uses setup_specs())
results <- run_specs(df = life_expec_dat,
y = c("life_expectancy"), # We add an additional dependent variable
x = c("gdp", "log(gdp)", "I(gdp^2)", "gdp:total_expenditure"), # We are not sure which independent variable is better
model = c("lm", "glm"), # We only use linear model or more complicated one
controls = c("population", "alcohol", "bmi"),
subsets = list(developed = unique(na.omit(life_expec_dat$developed))) # so it is 1 or 0, not sure why author of pkg choose to do it this way
)Third You inspect the specification curve to understand how robust your findings are to different analytical choices.
plot_decisiontree(results,
legend = TRUE)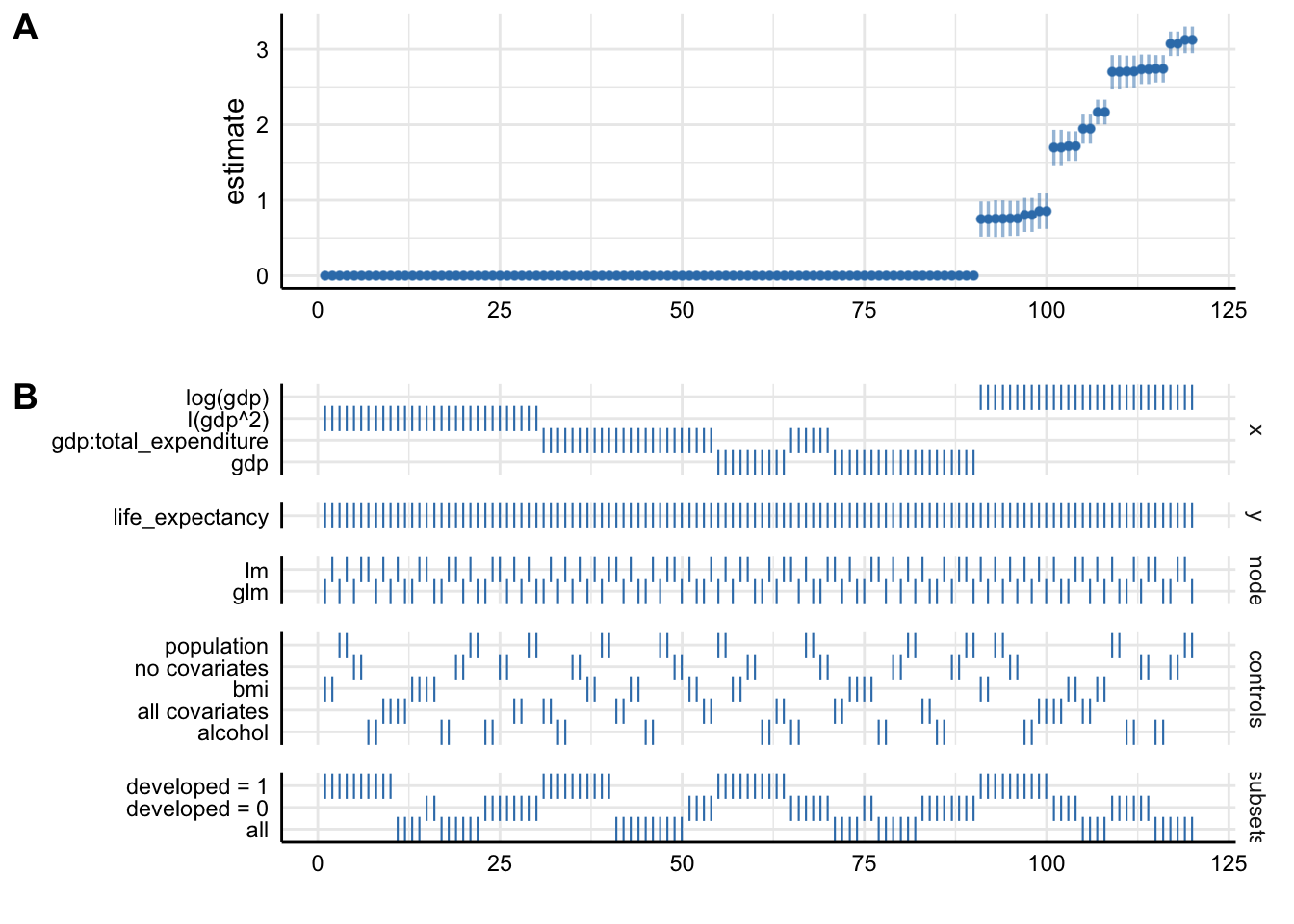 This decision tree helps illustrate where and when you had to make a
choice and how many models you might alternatively have specified.
This decision tree helps illustrate where and when you had to make a
choice and how many models you might alternatively have specified.
More concretely you could also look at some summary statistics for your p-values across the different model specifications:
summarise_specs(results,
var = c("std.error", "estimate"))The statistic mad stands for median absolute deviation.
A more intuitive way of inspecting the results might lie in visualising them.
plot_specs(results)
Plot A denotes: * X-axis = ordered model specifications (from most negative effect to most positive effect) * Y-axis = standardized regression coefficient for the IV-DV relationship * Points = the standardized regression coefficient for a specific models * Error bars = 95% confidence intervals around the point estimate
Plot B denotes: * X-axis = ordered model specifications (the same as panel A) * Y-axis (right) = analytic decision categories * Y-axis (left) = specific analytic decisions within each category * Lines = denote that a specific analytic decision was true for that model specification
Without putting much thought into our model it looks like gdp (depending on the specification) has either no effect or positive effects on life_expectancy. This just goes to show how careful one has to approach the task of correctly specifying a model!
Watch Out!
Be careful and selective when you use specification curve analysis! It is not a tool that you should apply to each and every potential paper. Especially, if you have not taken a course on causal inference. Specification curve analysis, when it includes variables that act as colliders or mediators might actually legitimise an otherwise wrongly specified model!
Model Output with broom 🧹
In the lecture we saw, that vanilla model lists can be unwieldy. Simon also pointed out that we are interested in three main aspects of out model outputs:
- Estimated coefficients and associated standard errors, T-statistics, p-values, confidence intervals
- Model summaries, including goodness of fit measures, information on model convergence, number of observations used
- Observation-level information that arises from the estimated model, such as fitted/predicted values, residuals, estimates of influence
Extracting this information from the vanilla model output is possible, but hardly ideal:
str(multiv_model_out[[3]])## List of 13
## $ coefficients : Named num [1:2] 64.763 0.955
## ..- attr(*, "names")= chr [1:2] "(Intercept)" "alcohol"
## $ residuals : Named num [1:2735] 0.227 -4.873 -4.873 -5.273 -5.573 ...
## ..- attr(*, "names")= chr [1:2735] "1" "2" "3" "4" ...
## $ effects : Named num [1:2735] -3617.34 202.2 -4.78 -5.18 -5.48 ...
## ..- attr(*, "names")= chr [1:2735] "(Intercept)" "alcohol" "" "" ...
## $ rank : int 2
## $ fitted.values: Named num [1:2735] 64.8 64.8 64.8 64.8 64.8 ...
## ..- attr(*, "names")= chr [1:2735] "1" "2" "3" "4" ...
....summary(multiv_model_out[[3]])##
## Call:
## .f(formula = .x[[i]], data = ..1)
##
## Residuals:
## Min 1Q Median 3Q Max
## -33.96 -4.72 1.62 6.41 20.76
##
## Coefficients:
## Estimate Std. Error t value Pr(>|t|)
....Tidying Model Objects
Luckily we don’t have to deal with this. We use a package called
broom that is part of the tidyverse and more precisely tidymodels. The latter offers a
lot of cool packages that have their own tutorials, so check them out!
💪.
We will only focus on broom. As a reminder, here are the three key broom functions that you need to learn:
tidy(): Summarizes information about model components.
broom::tidy(multiv_model_out[[3]], conf.int = TRUE, conf.level = 0.95)glance(): Reports information about the entire model.
broom::glance(multiv_model_out[[3]])augment(): Adds information about observations to a dataset.
augment(multiv_model_out[[3]], se_fit = TRUE) I mean, doesn’t this look a lot more accessible?
Broom with Many Models
Where broom really shines, is in dealing with many
models like we have in our multiv_model_out. It allows us
to easily move away from the generally painful-to-deal-with lists and
create yet retain each of the three important model returns. Since our
example replicates several variables in different models, I include the
.id argument in map. It adds a column to our tibble with an
model identifier. In our case this identifier consists
of the names of the elements in the list.
multiv_model_out_broom <- map_dfr(multiv_model_out[1:3], broom::tidy, .id = "model_type")
multiv_model_out_broomNow that we have our multiple model outputs in a format that we can
work with normally, it is time to start thinking about visualizing our
results. You can plot the residuals, the goodness of fit statistics etc.
Similarly you could also use the glance() to provide some
robustness statistics or descriptive tables.
What we will be focusing on in the next section is how to present your results!
Results with modelsummary 🥼
For this we will use the excellent modelsummary package
by Vincent
Arel-Bundock’s. Though there are many other options to choose from,
we highly recommend you give it a shot!
One reason that we like this package, is that it combines both ways of presenting model results, in a table or in a coefficient plot. As we saw in the lecture each method has its advantages and its drawbacks. Therefore, we will cover both here.
Using Regression Tables
The workhorse function in modelsummary is unsurprisingly
modelsummary(). 🤔
Here we only need to input the models list as, the function already takes care of the tidying for us.
modelsummary(multiv_model_out[[31]], output = "kableExtra")| Model 1 | |
|---|---|
| (Intercept) | 61.451 |
| (0.293) | |
| gdp | 0.000 |
| (0.000) | |
| percentage_expenditure | 0.000 |
| (0.000) | |
| alcohol | 0.394 |
| (0.034) | |
| bmi | 0.167 |
| (0.007) | |
| hiv_aids | −0.779 |
| (0.023) | |
| Num.Obs. | 2313 |
| R2 | 0.631 |
| R2 Adj. | 0.630 |
| AIC | 14787.7 |
| BIC | 14828.0 |
| Log.Lik. | −7386.873 |
| RMSE | 5.90 |
It looks pretty solid already, but unfortunately it is not yet good enough.
Set acceptable number of digits:
modelsummary(multiv_model_out[[31]],
output = "kableExtra",
fmt = "%.2f") # 2-digits and trailing 0And let us also report only the C.I. and get rid of the p-values and the Intercept term:
model_table <- modelsummary(multiv_model_out[[31]],
output = "kableExtra",
fmt = "%.2f", # 2-digits and trailing 0
estimate = "{estimate}",
statistic = "conf.int",
coef_omit = "Intercept") Would it not also look more professional if we had our coefficients begin with capital letter? And how about we get rid of some of the bloated goodness of fit statistics?
mod_table <- modelsummary(multiv_model_out[31],
output = "default",
fmt = "%.2f", # 2-digits and trailing 0
estimate = "{estimate}",
statistic = "conf.int",
coef_omit = "Intercept",
coef_rename=c("gdp"="Gdp",
"bmi"="Avg. BMI",
"alcohol" = "Alcohol Consum.",
"hiv_aids"= "HIV cases",
"percentage_expenditure" = "Health Expenditure (% of GDP)",
"life_expectancy" = "Life Expectancy"),
gof_omit = 'DF|Deviance|Log.Lik|AIC|BIC',
title = 'A Most Beautiful Regression Table')When using the kableExtra package you can even
post-process your table:
mod_table %>%
kable_styling(bootstrap_options = c("striped", "hover", "condensed", "responsive"), full_width = F, fixed_thead = T) %>%
row_spec(3, color = 'red') %>%
row_spec(5, background = 'lightblue')| Model 1 | |
|---|---|
| Gdp | 0.00 |
| [0.00, 0.00] | |
| Health Expenditure (% of GDP) | −0.00 |
| [−0.00, 0.00] | |
| Alcohol Consum. | 0.39 |
| [0.33, 0.46] | |
| Avg. BMI | 0.17 |
| [0.15, 0.18] | |
| HIV cases | −0.78 |
| [−0.82, −0.73] | |
| Num.Obs. | 2313 |
| R2 | 0.631 |
| R2 Adj. | 0.630 |
| RMSE | 5.90 |
Using Plots
If you are after an alternative way to present your results, you might also consider a nice coefficient plot 🎨
Again modelsummary provides a very accessible function
to do so: modelplot(). As with the regression table you
input the Model List and not the tidied
output. This already happens under the hood.
modelplot(multiv_model_out, coef_omit = 'Interc')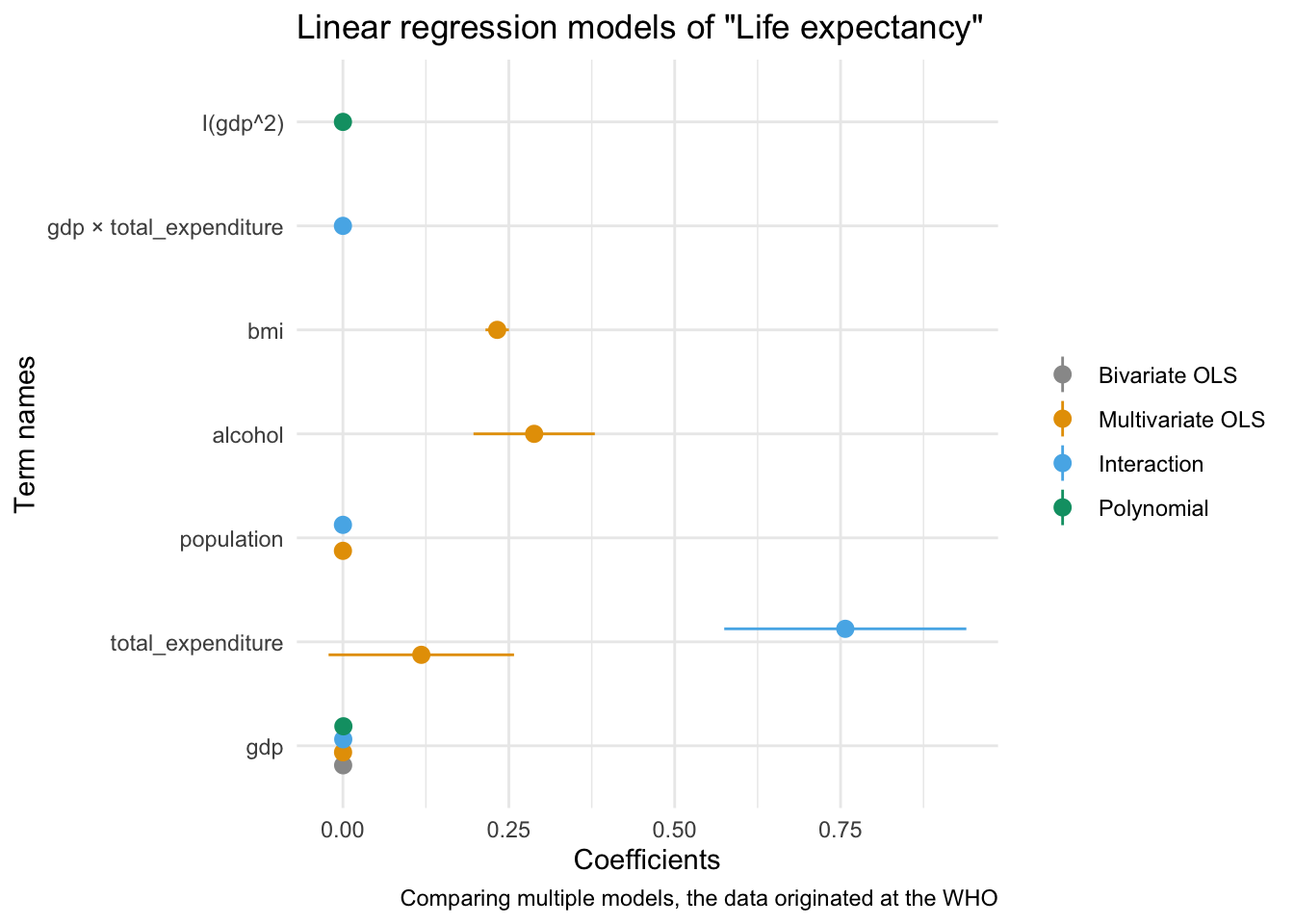
Now we only need to customise the plot to our liking:
modelplot(multiv_model_out, coef_omit = "Intercept") +
labs(x = 'Coefficients',
y = 'Term names',
title = 'Linear regression models of "Life expectancy"',
caption = "Comparing multiple models, the data originated at the WHO") 
We will cover the visualisation aspect in R in much greater detail in the coming session, so hopefuly the section on coefficient plot will become clearer then!
Exercises 🏋️
If you want to practice the above you can find more materials here.
Rather than do individual exercises this week we wanted to discuss different recording options for the upcoming Workshop Presentation.
Appendix 1: Workshop Prep 🎥
Soon the Workshop will be upon us. To help you prepare the recording of your presentation we wanted to present you with two options that
Using Zoom
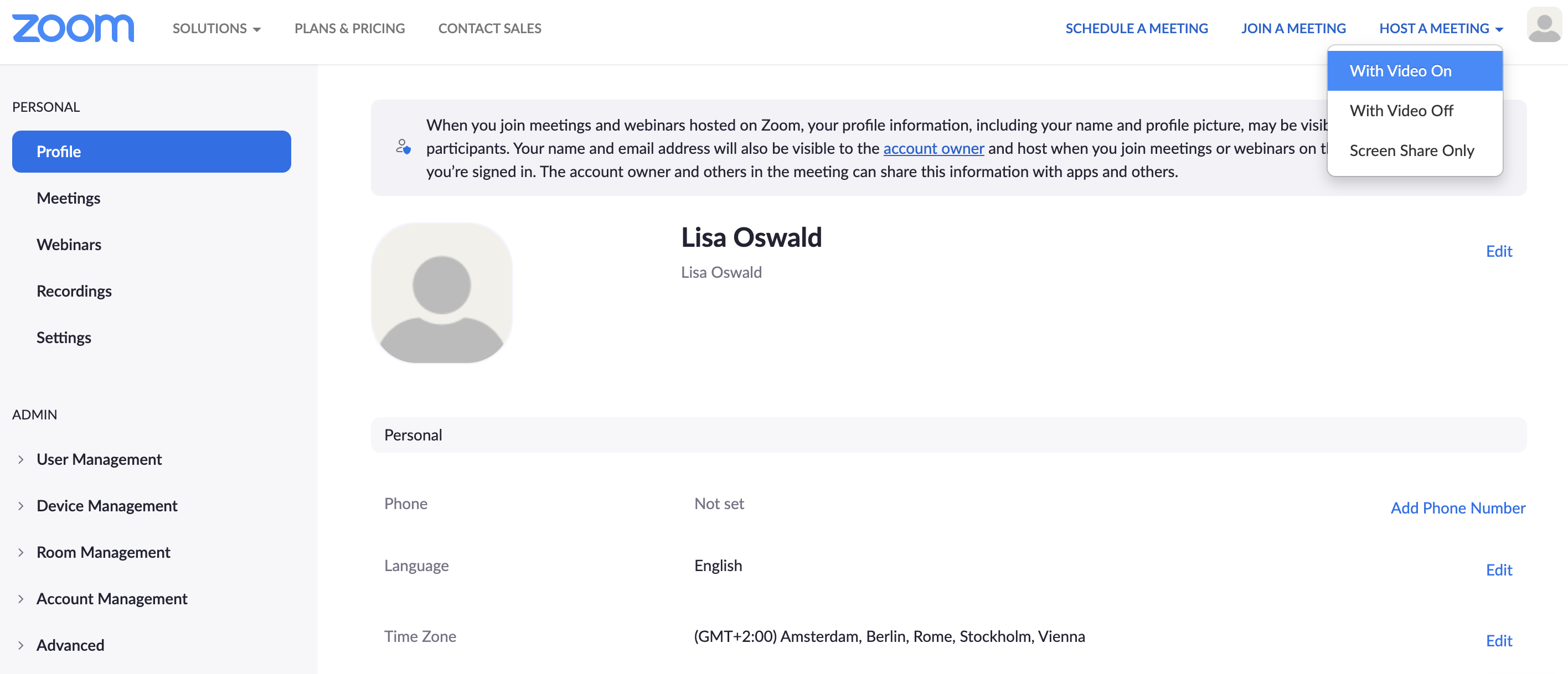
For this to work you need to register for a free Zoom Account here. In order to record yourself follow these steps:
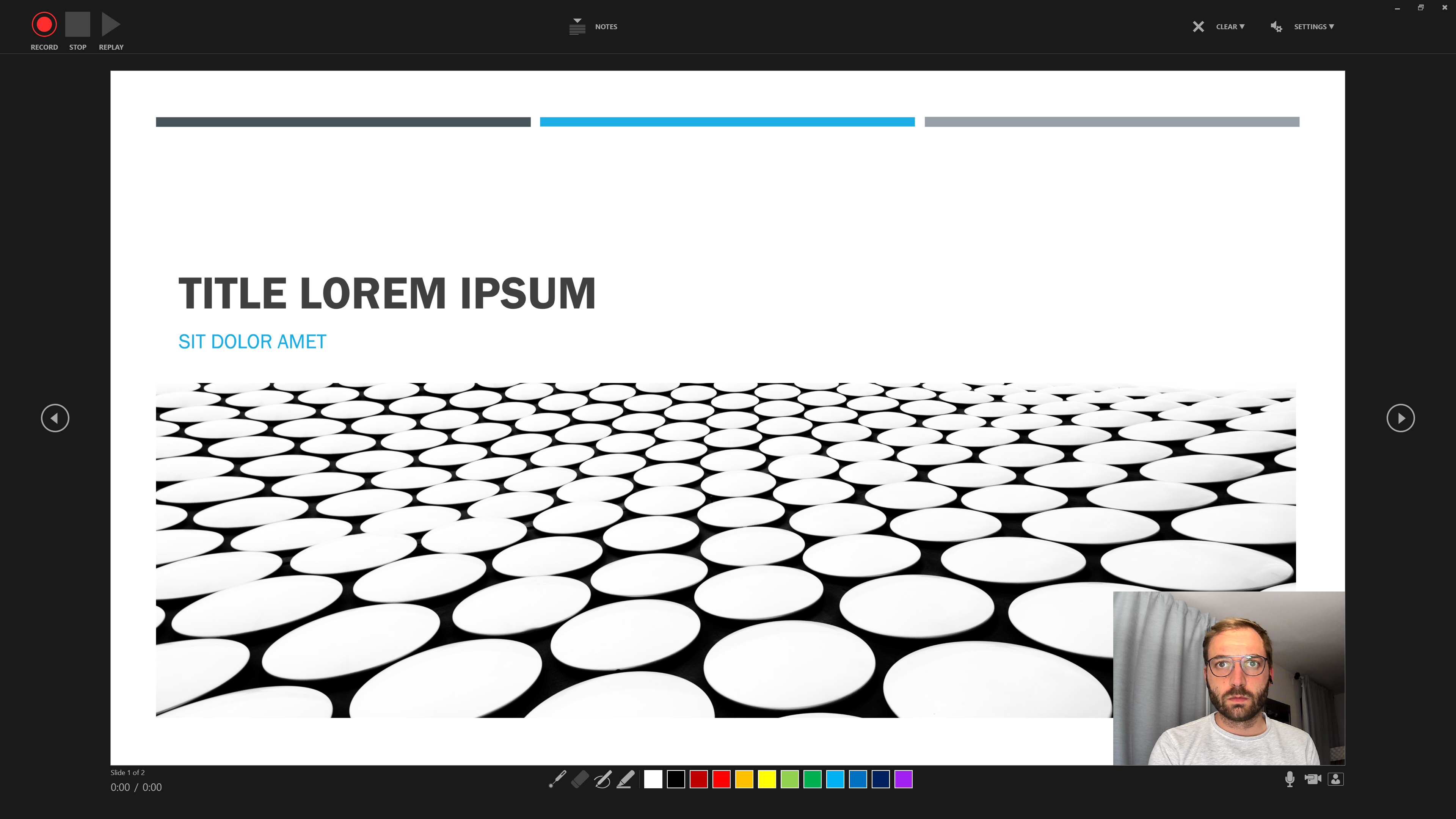
Step 1 Host a meeting by yourself and have the video turned on (it should be focused on you):
Step 2 Connect to audio and share screen (I usually chose Desktop 1, so I’m flexible to switch between applications - make sure to keep your desktop clean)
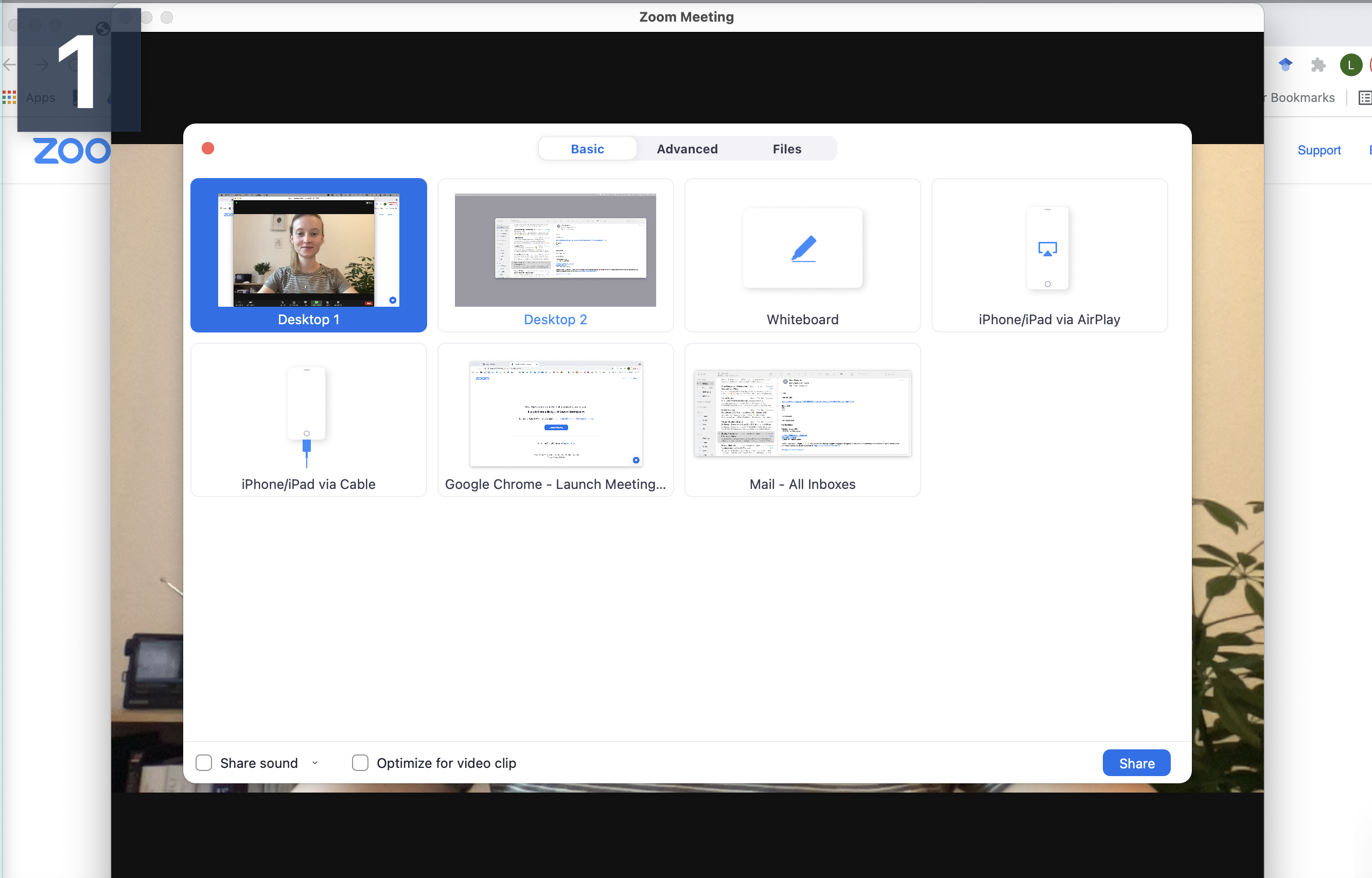
Step 3 Open the window/application that you will use for your presentation, place the little window with your face in one of the corners (so it does not hover somewhere distracting) and start recording.
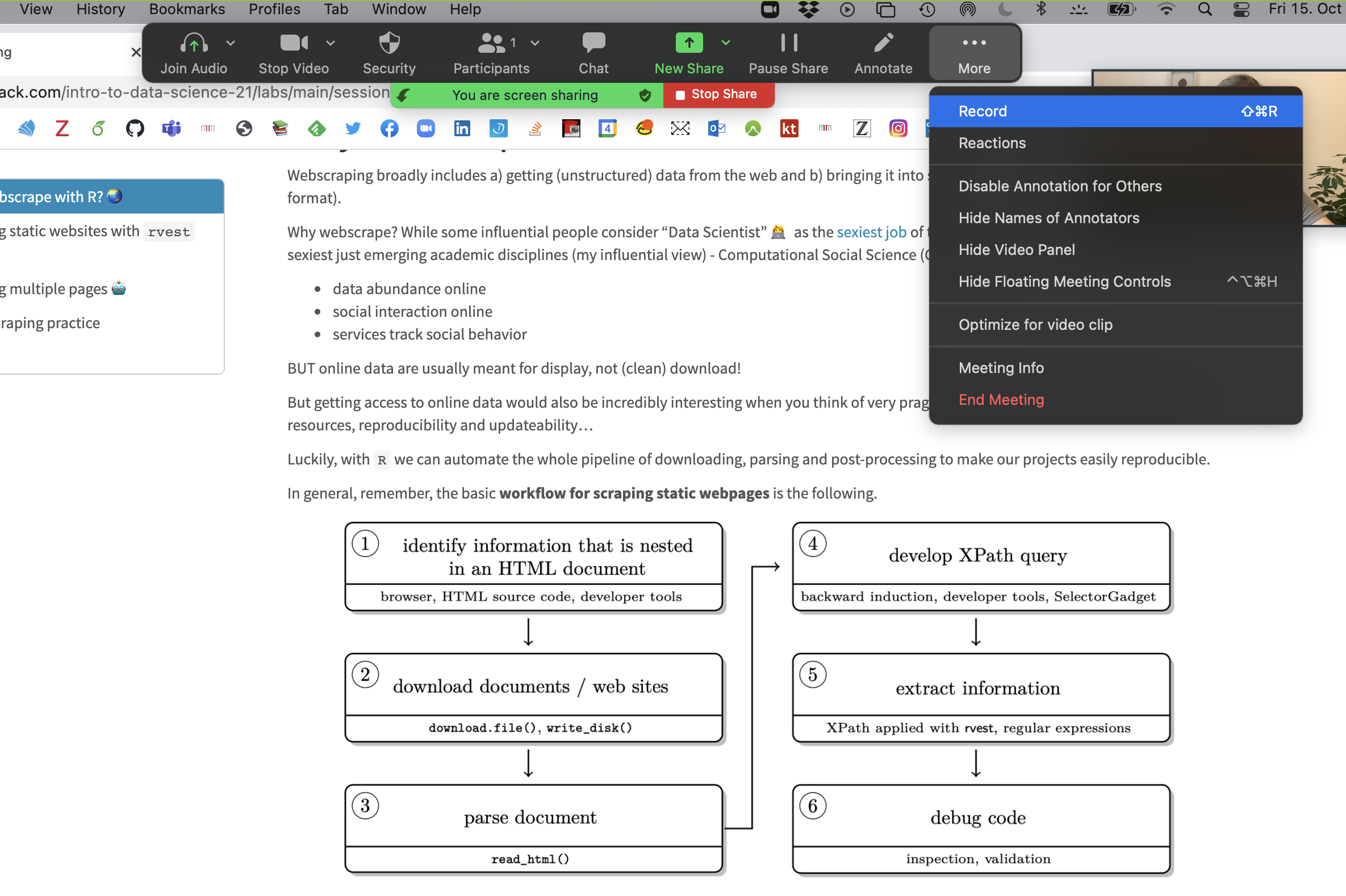
Now you have complete freedom to work with your laptop as usual (including doing a power point slide show), everything is recorded (except the zoom internal menu / dropdown options).
You can pause, continue (if necessary) and finally, stop the recording. The recording is saved and converted to mp4 after the end of the call.
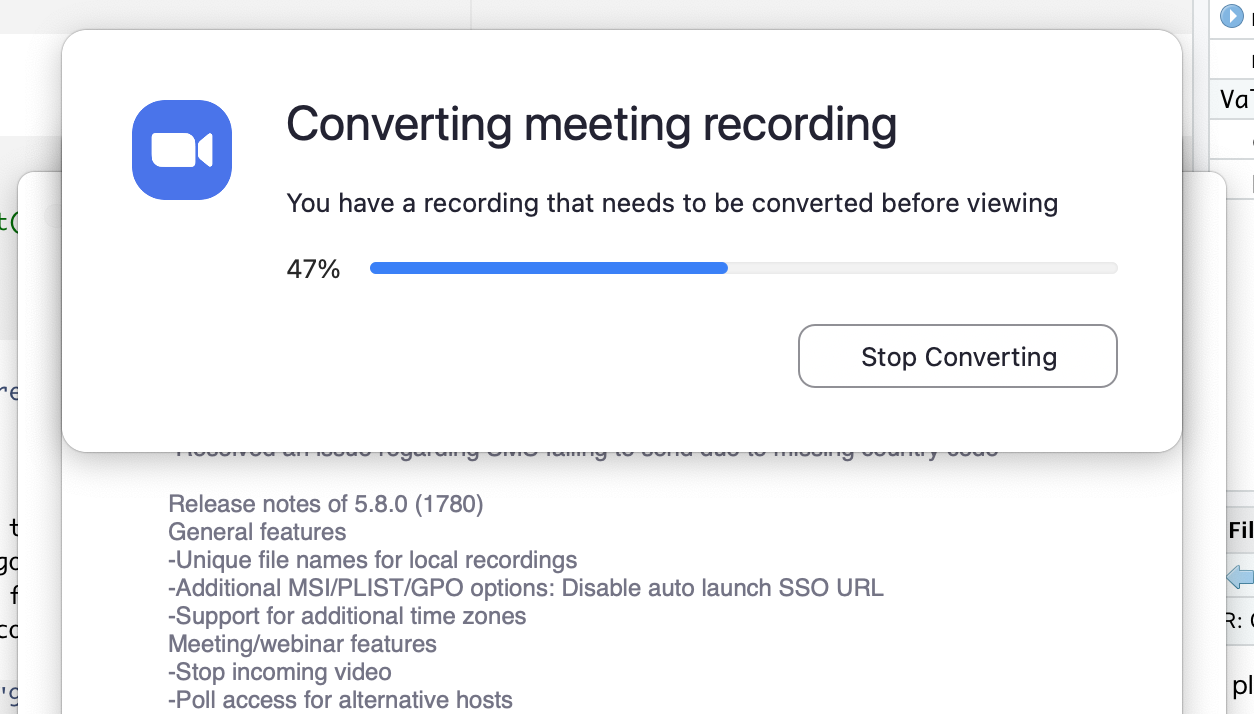
Should you feel the need to edit the video (for example, trim the length), the easiest option is to just use the tool inside the Quicktime player. This way you can edit the start and end of the video.
Using PowerPoint
For this to work you need to have an official MS Powerpoint licence and a finished presentation. In order to record yourself and the presentation follow these steps:
Step 1 Open a Presentation and click on Record Slide Show > Record from Current Slide (if you are at the start of the ppt.)
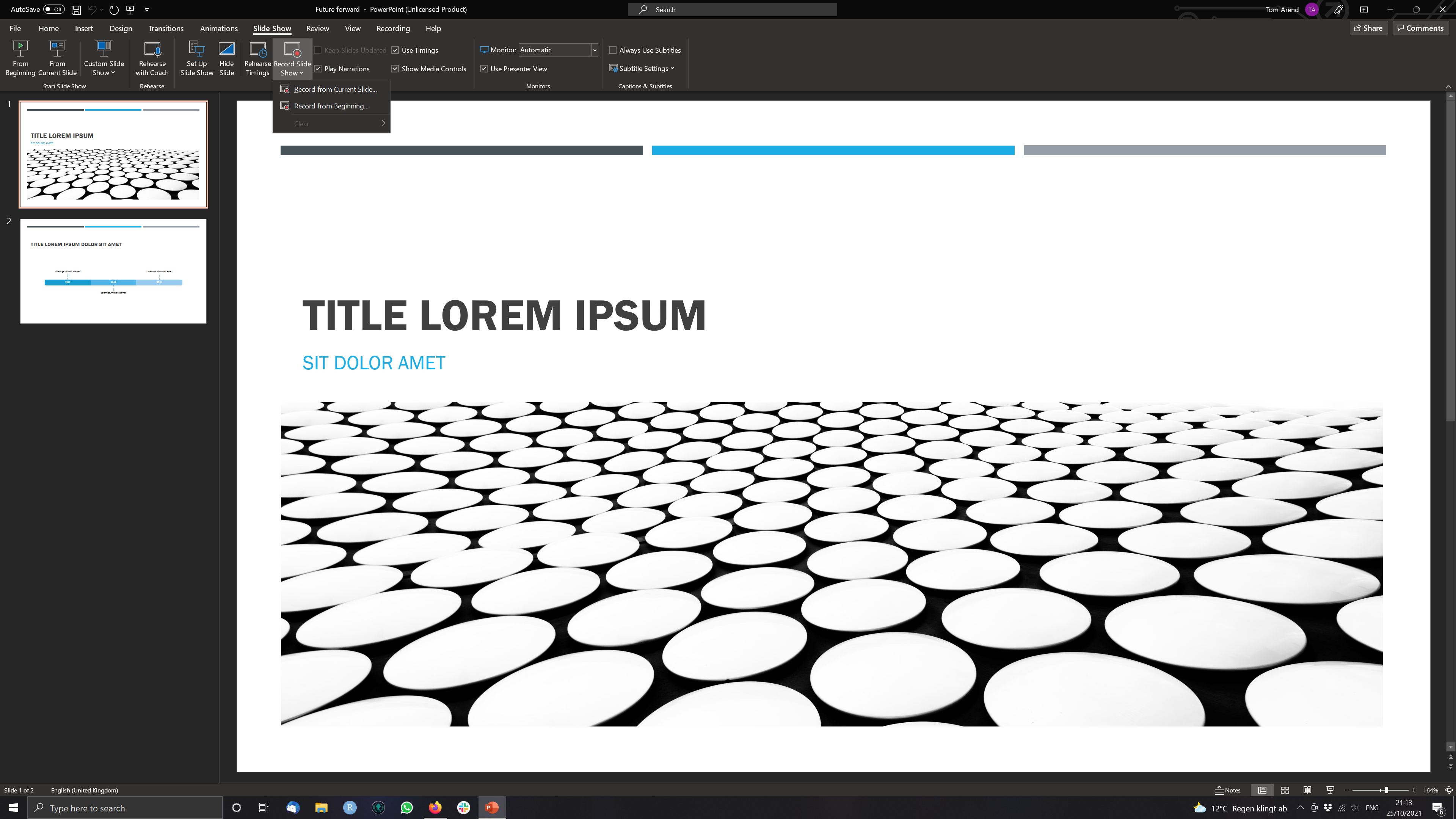
Step 2 Use the three buttons in the Upper Left to Corner to navigate the recording.
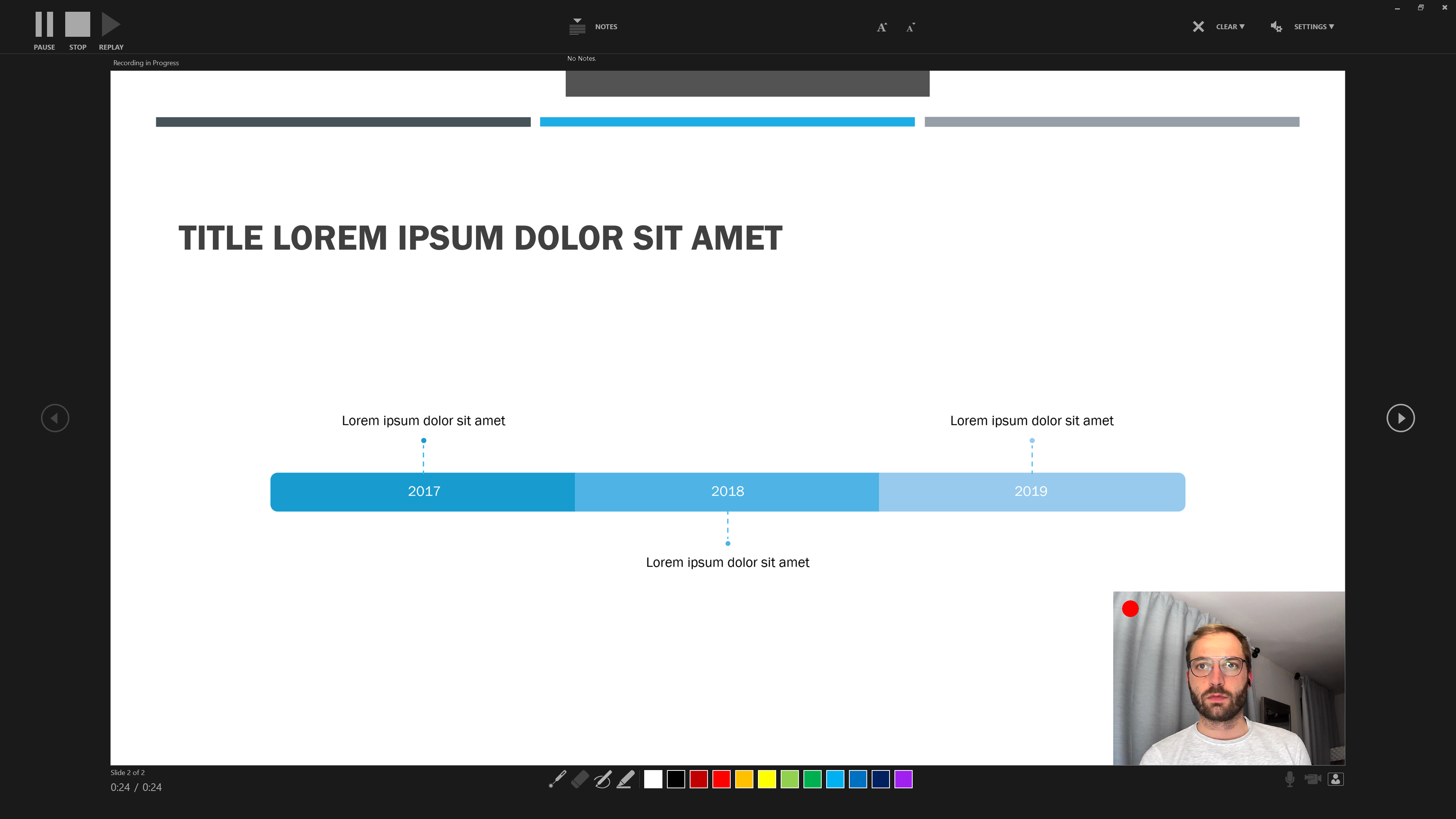
Step 3 Additional features include the option to use Slide Notes …
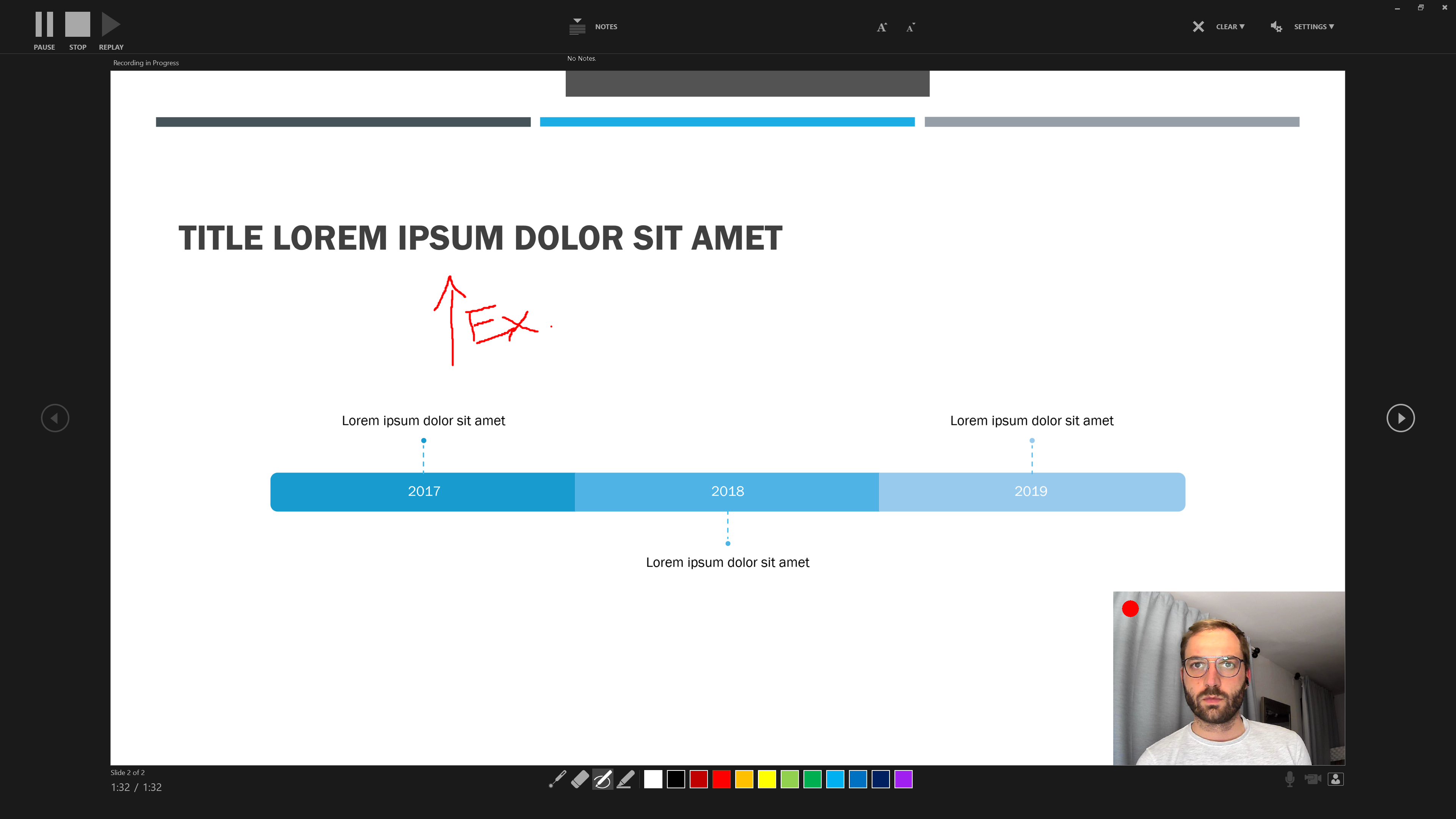
… and the colours and pens at the bottom to draw and highlight elements of a slide.
Step 4 Saving your recording as a video file
When you’re done recording, save your file and recording.
Then click on File > Export > Create a Video.
Powerpoint will now present you with a number of options regarding the quality of the video (go for the highest possible resolution) and whether you would like your video to include narration and slide timings. For most of you this should not apply.
Then choose a destination to save the video and you are all set.
Alternative Options to record presentations
The Readme on the Workshop Github contains a number of online video tutorials for a number of different Tools that you can have a look at. Similarly it offers more potent software tutorials on cutting and editing your videos.
Sources
This tutorial drew heavily on the vignette from the specr package as well as the Regression Section in McDermott’s Data Science for Economists by Grant McDermott.
A work by Lisa Oswald & Tom Arend
Prepared for Intro to Data Science, taught by Simon Munzert